https://www.youtube.com/watch?v=8bZUpnxd92U
The Microbiome signature of endometriosis: From dysbiosis to metallomics to targeted interventions
At the recent 11th Beneficial Microbes Conference, one presentation in particular sparked an unexpected misunderstanding: mine. The audience arrived expecting a methodical rundown of clinical interventions or perhaps a personal recovery protocol to combat disease. Instead, what they got was something quite different: a systems-level framework for understanding disease, presented through the case study of endometriosis (expanded step-by-step in the triangulation walkthrough). Many attendees, conditioned by academic routines to anticipate lists of “what to do,” unfortunately became disengaged when the talk focused on “how to think.” This mismatch between expectation and content led some to dismiss the talk prematurely, illustrating a broader issue in scientific culture. Scientists often crave concrete protocols and statistically validated checklists, but translational progress sometimes comes from reframing the problem entirely. In this case, the paradigm shift being offered was lost on those looking for familiar formats.
Although I did touch on the fact that I personally had resolved multiple chronic conditions over a decade ago, I also made it clear that the talk was not about my personal recovery methods or my n=1 experiments. It was about the thought process that led to integrative insights in research. Yet, because it didn’t align with the conventional “here’s a proven intervention” template, the real message didn’t land for much of the audience. This scenario invites reflection: How often do our expectations and academic conditioning blind us to new ways of thinking? And how can we avoid missing the value of a paradigm-shifting framework just because it isn’t packaged as a traditional protocol?
The Expectation: Protocols and Quick Fixes
To understand the misunderstanding, consider the typical mindset at scientific conferences. Researchers often anticipate tangible takeaways – e.g., a list of probiotic strains to use, a new dietary supplement, or a p-value backed recommendation to implement in practice. In the case of this talk, the title hinted at “targeted interventions,” and many likely assumed they’d receive a neat menu of treatment options. After all, much of academia trains us to seek discrete variables and linear solutions. We are comfortable with reductionist science: isolate one factor, test one hypothesis, present one list of significant findings. Over time, this creates a sort of protocol mindset – a belief that progress comes from accumulating specific answers rather than new ways of asking questions.
Such conditioning is reinforced by how we’re taught to evaluate information. An audience of microbiome scientists might expect to hear about specific bacterial species that could be modified to treat endometriosis, or a statistically significant clinical trial of a probiotic. An interdisciplinary, hypothesis-generating talk can feel “unsolidified” by comparison. Academic culture often prizes certainty and clear outcomes; anything that looks like a conceptual framework or a cross-disciplinary model may be perceived as too theoretical or not immediately actionable. In short, many were looking for “What should I do on Monday in my lab or clinic?” and when the talk instead asked them to reconsider how they think about a disease, many admitted to checking out.
The Reality: A Systems-Level Framework for Endometriosis
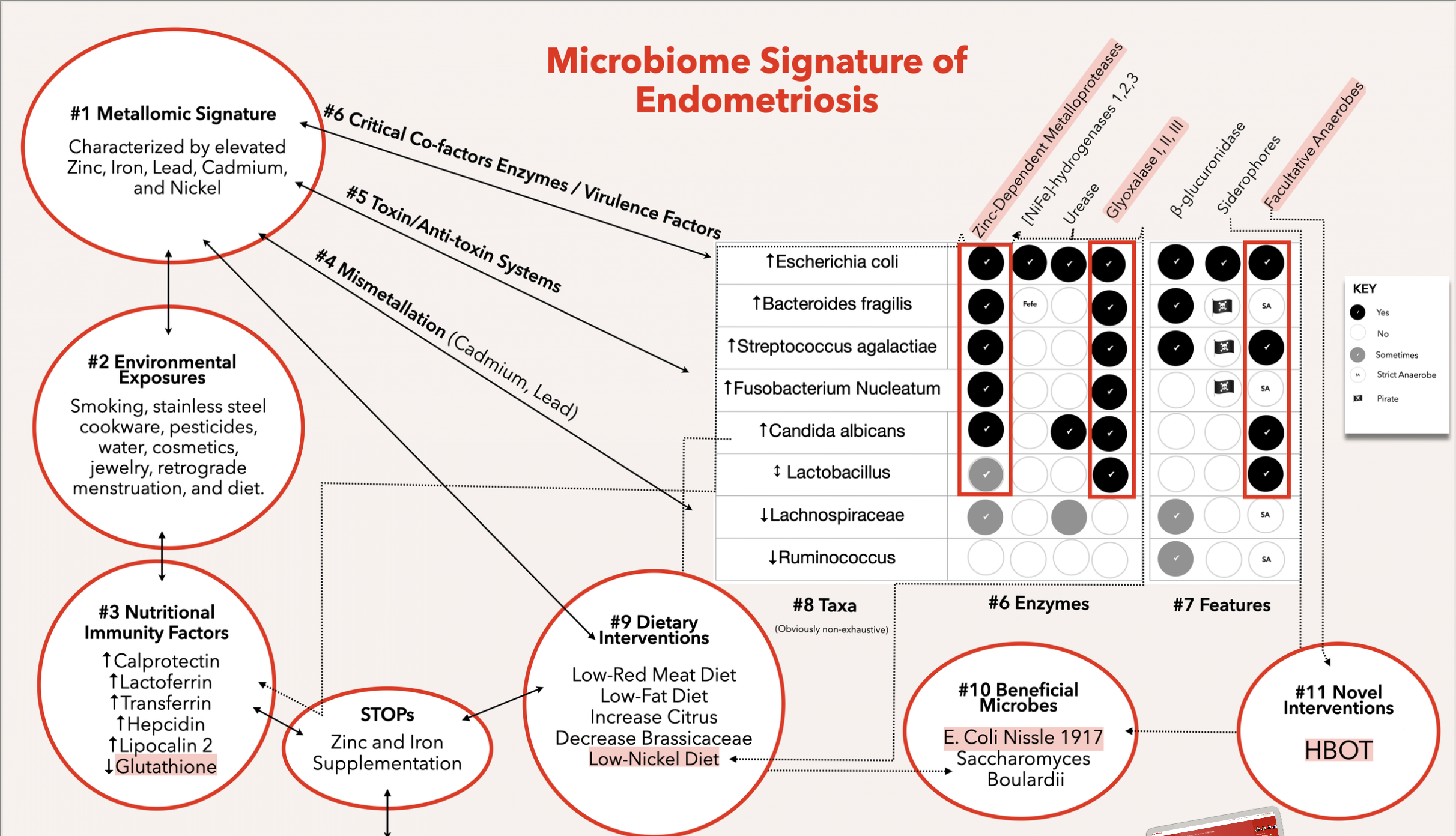
What I actually presented was a systems-level integration of multiple research domains, centered on a visual “Microbiome Signature of Endometriosis” diagram. Each bubble in that diagram represented a different disciplinary domain – environmental toxicology, microbiome signatures findings, metallomic signatures findings, microbial metallomics, immunology, estrobolomics and metabolomics, and microbial ecology – all interconnected as a scaffold for generating hypotheses, building disease models, and designing interventions. In essence, the presentation laid out a map of how to approach a complex disease like endometriosis from many angles at once. Over 70 studies were necessary to create a single slide, spanning everything from endocrine-disrupting chemicals to gut bacteria to immune dysfunction, all made accessible via a handout and the website microbiomesignatures.com/endometriosis. Rather than spoon-feeding a protocol, my aim was to implore the audience to see the bigger picture – to appreciate how disparate findings actually fit together into a cohesive framework.
Crucially, this framework was not a random collection of ideas; it was an evidence-based synthesis.Together, they illustrated why endometriosis should be seen as a systemic, multifactorial condition – not just a gynecological anomaly. In fact, a 2025 review reinforces this view, calling endometriosis a “chronic, inflammatory, and heterogeneous condition” with systemic impacts, not solely a localized gynecological disease 1. By visualizing these connections, the framework demonstrated how a change in one domain (say, environmental toxin exposure or a heavy metal burden) could cascade through microbial communities, metabolic pathways, and immune responses to influence endometriosis outcomes.
Consider the metallomics bubble in the diagram. Not many endometriosis talks would mention metallomics – the role of trace metals – but here it was front and center. Why? By incorporating metallomics into the framework, I could illustrate a truly interdisciplinary scaffold: one in which a gynecologist, a microbiologist, an immunologist, and an environmental health scientist would all see their respective pieces of the puzzle, as well as how those pieces connect.
Each domain bubble – be it Environmental Toxicants, Gut/Vaginal Microbiome, Trace Metals, Immune Dysfunction, or Microbial Ecology – was supported by specific studies. More importantly, the interdisciplinary intersections between those bubbles are where new hypotheses bloom. The Microbiome Signature of Endometriosis diagram serves as a hypothesis-generating engine. It states: Given all these factors, what combinations could drive disease in a particular patient? How might we intervene in one domain to influence another? Can information from one domain identify gaps in knowledge, or mistakes being made in another? This kind of thinking is inherently translational – it’s building a bridge from scattered research findings toward integrated clinical strategies. It’s the opposite of a reductionist, taxon-centric approach. And that was precisely the point of the talk.
“How to Think” vs “What to Do”
One of the key messages was that the real value lies in demonstrating how to approach complex problems, not prescribing exactly what to think or do in every scenario— because those prescriptions effectively squander creative intelligence.
This was a talk about mindset and methodology more than a list of results. I briefly referenced my personal health journey – resolving multiple chronic conditions by following evidence across disciplines – as an existence proof that there was real power in this systematic thinking approach. How else could I have had such drastic improvements, when modern medicine doesn’t see this kind of resolution?
Nonetheless, I did not explicitly share my personal regimen or a cookbook for others to copy. I did not talk about the patients or clinicians I've worked with, or the conditions we have successfully treated. Why? Because the aim was to empower researchers and clinicians to develop their own integrative insights, not to hand them a prefab solution. And admittedly, I see microbial metallomics and metallomic signatures sorely missing in the literature. It was such a crucial discovery, and it needed to be added to the "omics" mix. At the very least, I think while some messaging was lost, this one landed well.
Still, this distinction between how and what is critical in science. Teaching someone what to think yields a finite answer (which might become obsolete or may not generalize). Teaching someone how to think equips them with a process to find new answers endlessly. The talk provided a working example of how to think in an interdisciplinary, systems-level way: how to gather clues from many fields, how to spot patterns, and how to assemble those into testable models of disease. For many in the audience, used to conference talks that conclude with, “And thus, treatment X significantly reduced symptom Y,” this approach felt unconventional. Some may have even felt it was impractical.
But allow me to be clear—it is exactly this thought process that drives innovation. It’s the same process behind notable scientific breakthroughs, where old models were transcended rather than tweaked.
Unfortunately, those who tuned out early missed the fact that this framework did, in fact, point toward interventions – just not in the (perhaps simplistic) way they expected. The final portion of the presentation did touch on interventions, but framed within the system. For example, recognizing nickel’s role naturally suggests a low-nickel diet or chelation strategy; understanding the gut microbiome’s contribution opens up probiotic, prebiotic, or even fecal transplant considerations; identifying chronic inflammation loops suggests anti-inflammatory or immune-modulating approaches. Indeed, several validated and promising interventions for endometriosis align with these insights (from hyperbaric oxygen therapy to targeted antibiotics and diets56).
It should be stated, however, that I intentionally avoided just listing these without context. The interventions were presented as logical offshoots of the framework – examples of what thinking broadly can yield. The framework helps you know where to look. And the take-home lesson was not “use this probiotic or that drug”; it was “here’s how you can think more systematically to discover and vet interventions by understanding the whole system.”
That is a richer lesson, but also a less familiar one in a field used to incremental advances. I was afforded the benefit of not having my way of thinking taught out of me in an effort to maintain a narrow identity as a scientist— some of the best scientific advances in the world came from outsiders who simply “thought differently.”
Breaking Out of Reductionist Habits
The conference also highlighted another deeper cultural habit in science: reductionism. Reductionist, taxon-centric thinking in microbiome research, for instance, often focuses narrowly on “which bug causes X disease” or “which bug can we use to treat X disease.” Yet leading scientists are recognize that this approach can be limiting. A 2023 review in Pathogens bluntly pointed out that our “continued reliance on taxonomic indicators to define microbiomes” has become an impediment – it can confound analyses and cause us to miss true causal relationships 7. The authors advocate for moving away from a purely taxonomic approach and instead applying systems biology principles7. In simpler terms, focusing on cataloguing microbes like a bird-watcher (“which species are present or absent”) may not get us far in understanding diseases. What we need is to understand what microbes are doing (metabolically, immunologically) and how they interact with the host and environment as a system. This isn't outlandish, it's critical. And, this sentiment among other researchers aligns perfectly with the framework presented in the talk.
It was an invitation to abandon the comfort of reductionism and to embrace complexity. Granted, thinking in systems is harder than thinking in checklists. It requires tolerating ambiguity, crossing disciplinary boundaries, breaking down silos, and synthesizing large amounts of information. These are skills that traditional academic training doesn’t always emphasize. In fact, academic conditioning often teaches one to specialize deeply, to trust only the narrowest controlled experiments, and to be skeptical of anything that doesn’t fit neatly into established paradigms. While rigor and skepticism are pillars of science, they should not become blinders that prevent us from seeing the forest for the trees.
Consider how interdisciplinary thinking is becoming increasingly crucial in microbiome science. The human microbiome isn’t an isolated subject – it intertwines with diet, environment, neurology, endocrinology, etc. Some commentators have aptly noted that the microbiome is “not just a biology problem. It’s a systems problem,” requiring collaboration across many fields to truly understand and harness 8. Indeed, forward-looking initiatives in science encourage breaking silos. Multi-omics integration – combining data from genomics, metabolomics, metallomics, immunology, and more – is heralded as a pivotal approach to unravel complex interactions that single-discipline studies miss 9. Transdisciplinary research is no longer a luxury; it’s a necessity if we want to solve complex diseases.
Endometriosis is a prime example of a condition that cannot be fully understood through one lens. Hormones, immune responses, gut microbes, toxic chemicals, genetic factors – all contribute to its pathogenesis 310. If we cling to conventional epistemology – for instance, insisting that only double-blind trials in one domain are worthwhile, or that each field stays in its lane – we impede innovation. We often overlook phenomena that don’t fit our mental model. We risk dismissing bold hypotheses that might lead to breakthroughs. Is this good science?
The premature dismissal of the talk by some attendees is a perfect case-in-point. I dare say it's even a form of intellectual myopia: “This isn’t what I expected, this is unconventional, unorthodox, and uncomfortable, and therefore it must not be useful.” Not only is it unfortunate that many saw this as a n=1 presentation (and thus, sorely missed the point entirely), but even if it were, history is full of examples where disruptive ideas and n=1 experiments were initially ignored or ridiculed by the establishment, only to later revolutionize the field. To avoid repeating that pattern, scientists and clinicians must actively practice intellectual flexibility or risk intellectual stagnation.
This means listening to ideas outside our immediate expertise, evaluating frameworks on their merits (even if they don’t present a tidy dataset yet), and being willing to update our mental models if evidence calls for that. As one theory in the talk suggested, even something as outside-the-box as environmental toxin exposure can be integral to a disease like endometriosis – implying that a clinician might need to ask her patient about chemical exposures, diet, and heavy metals, not just family history and hormones11. That’s a broadening of scope, and it might feel uncomfortable to those who are traditionally trained. Yet, it could open the door to interventions that conventional thinking never considered. It is why outsiders offer such an advantage for clinical breakthrough.
Embracing Systems Thinking for Translation to Clinic
Ultimately, the purpose of the talk – and the purpose of this reflective post – is to champion systems-level thinking as a cornerstone for translational research. That’s it. That’s the method you were wanting. Like I said in my talk, I gathered all of the data I could get my hands on to come up with a more coherent idea of what sort of dysbiosis I was experiencing. I made associations, and I evaluated strategies within that framework to decreased pathogens and increase beneficial taxa. I said that, and then I showed what that sort of interdisciplinary systems-level thinking looked like applied to endometriosis.
And most importantly, translational science is all about turning knowledge into real-world applications, bridging the bench to the bedside. For the microbiome field (and biomedical science in general) to achieve meaningful clinical breakthroughs, we must venture beyond business as usual. Sticking to comfortable methods and siloed perspectives will only perpetuate stagnation. As the conference talk highlighted, there is an urgent need for breakthroughs in challenging diseases like endometriosis, where current treatments are suboptimal and no definitive cure exists. Breakthroughs rarely come from doing the same old thing slightly better; they come from seeing the problem in a new light.
Question
How many people stopped to ask themselves, despite my litany of devastating diseases, how I managed to resolve such harrowing, and often fatal conditions? Perhaps a clinician would have more intuitively understood that I would not be alive if I didn't approach my condition differently. Or, at the very least, I would still be suffering from these debilitating, chronic conditions. That lack of curiosity says more about some in the audience than it says about me. Is a lack of curiosity, in the face of something so surprising, good science?
The integrative framework presented – using endometriosis as an example – is exactly about shining that new light. It suggests that by creating a comprehensive map of a disease (one that includes microbes, metals, molecules, and more), researchers can identify points of intervention that might otherwise be missed. It offers a scaffold for hypothesis generation: for instance, if heavy metal exposure and gut dysbiosis both appear important, one might hypothesize that certain metal-chelating therapies (like DMG) or microbiome-targeted diets could help patients (indeed, the Low-Nickel Diet and use of lactoferrin for iron modulation were mentioned as validated interventions 5, 6, emerging directly from such hypotheses). Likewise, the framework aids in clinical modeling, helping to conceptualize why a patient might experience both gut symptoms and pelvic pain, or why an immune-modulating drug might alleviate what was previously thought to be a strictly hormonal disease. This broad view is empowering. Instead of waiting passively for a randomized trial to provide a single answer, scientists equipped with a framework can proactively design experiments and intervention trials that account for multiple dimensions.
It’s important to underscore that showing how to think systemically does not mean abandoning scientific rigor. On the contrary, it provides a more robust context within which rigorous experiments can be designed. For example, a reductionist approach might test one probiotic for endometriosis and obtain a lukewarm result, leading researchers to dismiss it. A generalist, or natural systems thinker, might realize that the probiotic’s efficacy could depend on other factors, such as the patient’s heavy metal load, or immune status – factors not accounted for in a narrow trial. This is precisely what the literature warns against in the dangers of causal reductionism in microbiome science. Thus, the next study can be refined to stratify patients by those variables, potentially revealing a clear benefit in the right subgroup. In this way, the interdisciplinary framework guides more effective and nuanced research. It’s not opposed to statistical validation; it actually helps ensure that what gets tested has a strong rationale grounded in a mechanisms-driven, holistic understanding of the disease.
Conclusion: Stay Curious, Stay Open
The fundamental misunderstanding at the conference – expecting a list of “what to think” instead of learning “how I think” – turned out to be a learning moment. It shone a light on how our expectations, shaped by academic convention, can close our minds at the very moments when we should be most open. As scientists and researchers, we pride ourselves on curiosity and objectivity. Yet, when faced with an unfamiliar type of presentation, some of us reacted by shutting down rather than engaging. To truly push microbiome science (and medicine at large) forward, we must cultivate the opposite reaction: lean in to new frameworks, especially when they challenge our assumptions in a surprising way.
Endometriosis was the case study here, but the message transcends any single disease. The call to action is for intellectual flexibility and systems-level curiosity. We should ask ourselves: Are we willing to venture beyond our comfort zones of data and theory? Will we acknowledge that complex problems demand multi-dimensional solutions? The next time a colleague presents a paradigm-shifting idea or thought process that doesn’t fit the usual mold, perhaps resist the urge to dismiss it. Instead, examine it, ask critical questions, and consider how it might fill the gaps left by conventional approaches.
Sidebar
The framework I offered could mechanistically answer several outstanding questions left by other endometriosis researchers.
In the end, the framework wasn’t about telling anyone what to think about endometriosis – it was about demonstrating a way of thinking that could be applied to any complex biomedical challenge. Those who recognized this found the talk invigorating. They saw a scaffold for innovation rather than a deviation from the norm. And that is the take-home lesson for all of us: the real value in science is often in the how.
Mastering the how – how to integrate disciplines, how to generate hypotheses from scattered clues, how to remain open to the unexpected – is what will allow microbiome research to blossom into meaningful clinical applications. We owe it to ourselves, and to the patients who stand to benefit, to embrace such paradigm shifts. Let’s stay curious, stay open, and welcome the frameworks that show us new ways to think. They might just offer the blueprints of tomorrow’s breakthroughs.